For decades, the pharmaceutical industry sought single drugs for single diseases. But in recent years, the focus has changed to treating interconnected biological systems instead of just isolated symptoms. Metabolic health, neurodegeneration, cardiovascular disease, and inflammation are increasingly understood as overlapping networks rather than independent silos.
Glucagon-like peptide-1 receptor agonists (GLP-1RAs), has emerged as a powerful example of this transition. Originally developed to regulate blood glucose level in patients with type 2 diabetes, these therapies have now gained significant attention for their role in weight management and broader metabolic health. While most mainstream media describe GLP-1 as a "satiety hormone", This comparison is similar to calling the internet a messaging app
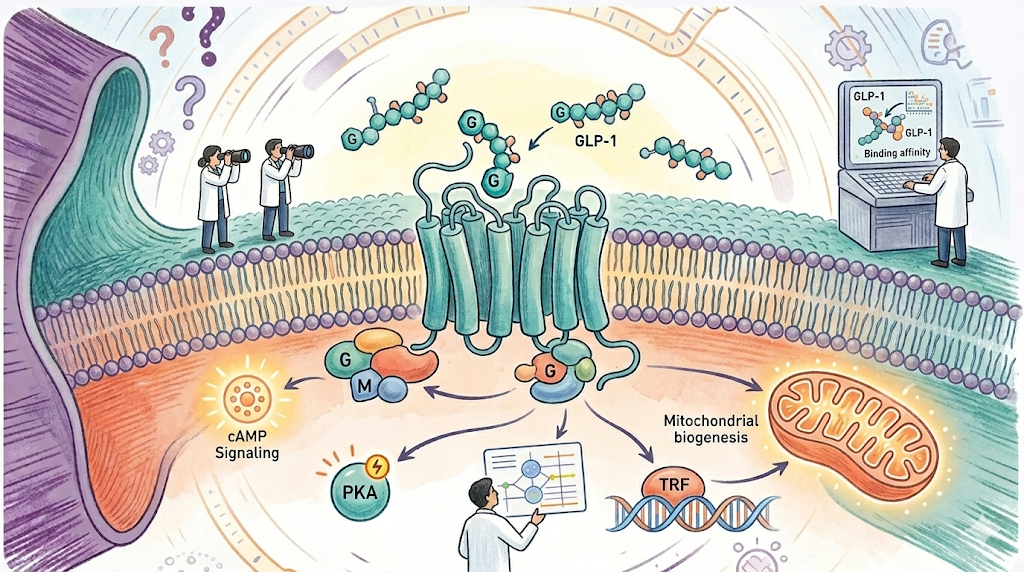
According to the Patel & Niazi (2025) review, emerging research suggests that the strength of GLP-1 receptor signalling lies in mitochondrial health and autophagy. When activated, it doesn't just signal the brain to stop eating but starts a cellular clean up process by changing how genes related to insulin sensitivity and lipid metabolism are transcribed. It improves mitochondrial biogenesis, which boosts function and lowers oxidative stress which results in cleaner energy production at the cellular level, with less metabolic waste harming nearby tissues. The pathway starts with the L-cells in the gut, which produce GLP-1, which then moves through neural circuits in the hypothalamus that manage hunger and energy balance. The gut-brain-mitochondria axis redefines metabolic regulation, evolving our understanding from a binary appetite switch to a complex architecture of cellular quality control.
The therapeutic reach of GLP-1 RAs is currently undergoing an "Indication Land Grab." As clinical data accumulates, these molecules are being explored across multiple organ systems, driven by their ability to modulate inflammation, mitochondrial efficiency, and metabolic signaling.
What took decades with semaglutide now happens in months. AI has compressed the path from target discovery to clinical candidates by screening billions of molecules in silico, estimating binding, safety risks, and pharmacokinetics before anything reaches the lab.
But the story is not perfect. The biggest concern is sarcopenia. Around 25 to 40 percent of weight loss can come from lean muscle, raising long term health questions. That is why the next investment wave is not just more GLP-1 drugs but GLP-1 plus adjuvants, especially myostatin inhibitors aimed at preserving muscle.