Disease Biology
Disease:
Chemotherapy-Induced Nausea and Vomiting (CINV)
Prevalence:
Affects up to 70–80% of patients receiving highly emetogenic chemotherapy without optimal prophylaxis
Disease Mechanism:
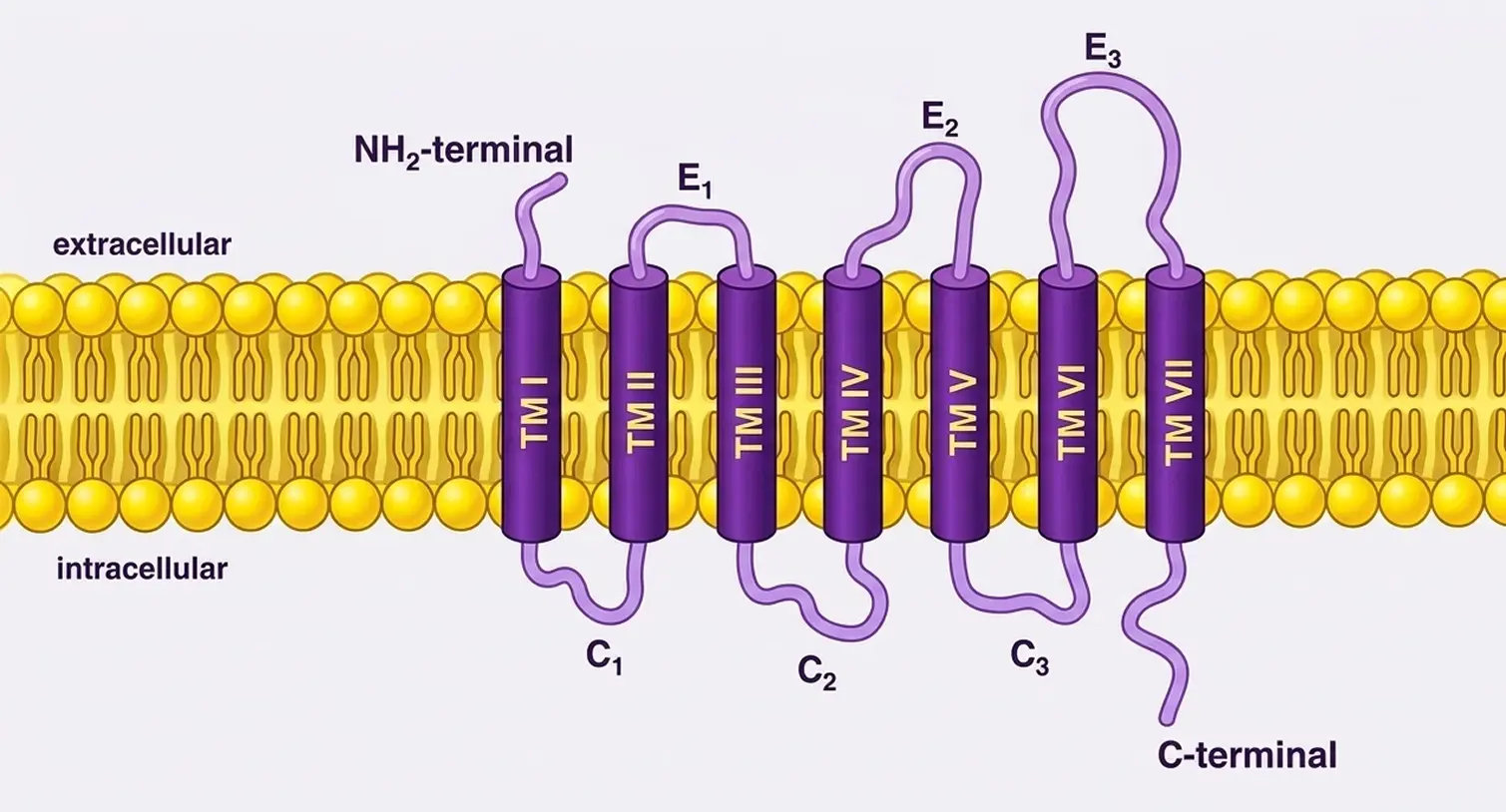
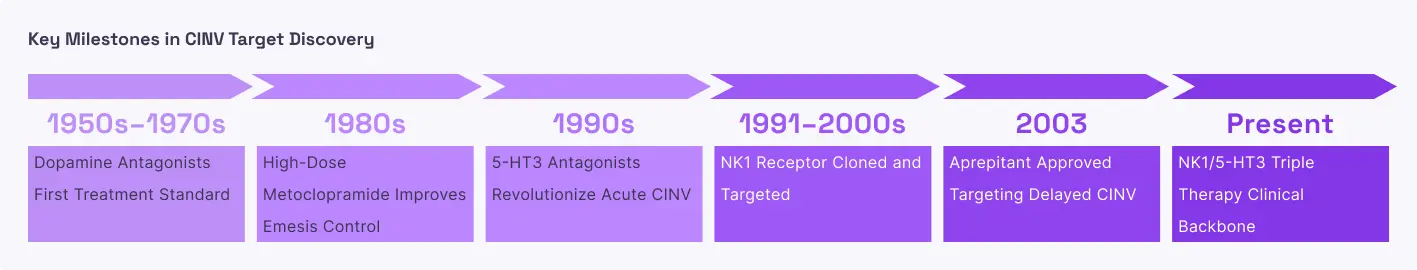
CINV is a complex defense mechanism where the body mistakenly identifies chemotherapy drugs as life-threatening toxins that must be expelled. The process occurs in two phases: the acute phase (within 24 hours) and the delayed phase (24 to 120 hours). While the acute phase is largely driven by serotonin, the delayed phase is predominantly controlled by the release of Substance P from the vagus nerve and central nervous system. As chemotherapy circulates, it triggers the release of Substance P, which then travels to the brainstem and binds to NK-1 receptors. This activation coordinates a massive muscular and neural response across the gastrointestinal tract and the brain, leading to persistent, debilitating vomiting that can cause dehydration, malnutrition, and even force patients to discontinue life-saving cancer treatments.
Why Target the drug target is used in this disease?
NK-1 receptor antagonists are uniquely effective for the delayed phase of CINV, where other common anti-nausea drugs (like serotonin blockers) often fail. By occupying the NK-1 receptor, these drugs act like a "lock-protector," preventing Substance P from fitting into the receptor and triggering the emetic cascade. Because these drugs can cross the blood-brain barrier, they provide a "central" blockade directly in the brain’s vomiting center. This approach has transformed cancer care, allowing patients to maintain their nutritional status and complete their full course of chemotherapy with significantly less distress than was possible in previous decades.
Disease:
Chronic Pruritus (Chronic Itch)
Prevalence:
Approximately 98 million prevalent cases across major global markets in 2024.
Disease Mechanism:
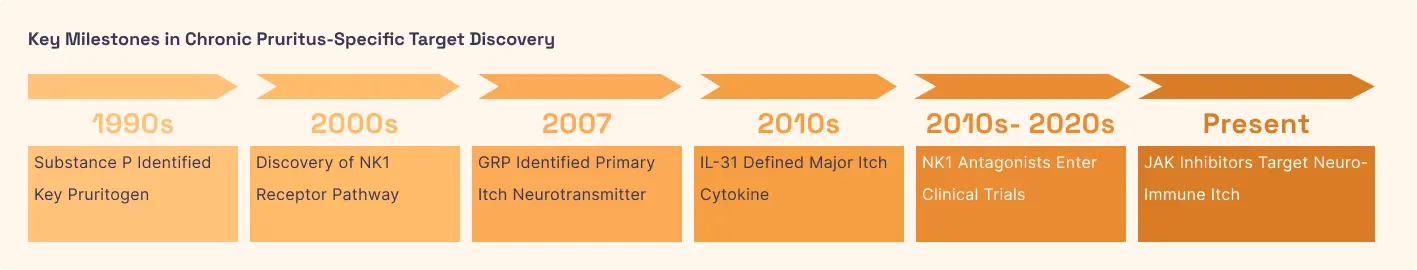
Chronic pruritus is more than just a surface-level itch; it is a neurological dysfunction where the skin's "itch-sensing" nerves become hyper-sensitized. In conditions like atopic dermatitis or kidney disease, skin cells and immune cells release an excess of Substance P. This molecule binds to NK-1 receptors located on the ends of sensory nerves and on skin cells called keratinocytes. This creates a "vicious cycle": the activation of NK-1 receptors causes the release of inflammatory chemicals, which further irritates the nerves and leads to more itching and scratching. Over time, this constant stimulation can cause the nerves to rewire themselves, making the sensation of itching feel permanent and intensely painful.
Why Target the drug target is used in this disease?
Targeting the NK-1 receptor in the skin and spinal cord provides a way to "mute" the itch signal at its source. NK-1 antagonists block the interaction between Substance P and the sensory nerves, effectively quieting the overactive signaling. This is particularly valuable for "recalcitrant" itch cases that do not respond to standard antihistamines because the underlying cause is neurological rather than just an allergic reaction. By breaking the itch-scratch cycle, these therapies allow the skin to heal and significantly improve the sleep quality and mental health of patients suffering from long-term dermatological conditions.