Disease Biology
Disease:
Obesity
Prevalence:
Metabolic & Cardiometabolic Disorders
Disease Mechanism:
Obesity is a complex, chronic disease characterized by excessive fat accumulation that impairs health due to a biological dysfunction in the way the brain and gut communicate about energy storage . In many people with obesity, the brain’s "set point" for weight is recalibrated higher. The hormones that should signal fullness are often under-produced or ignored by the brain, while hunger signals remain overactive. This creates a cycle where the body fights to maintain a higher weight, leading to systemic inflammation and a significantly increased risk for heart disease and various cancers .
Why Target the drug target is used in this disease?
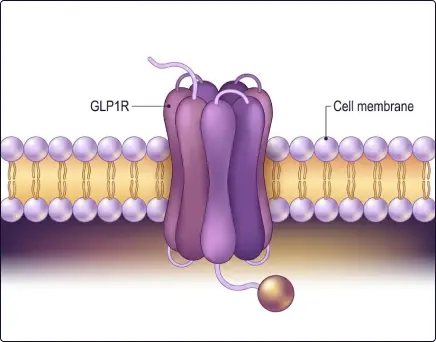
Targeting the GLP-1 receptor directly fixes the broken communication between the gut and the brain. By using a drug that mimics natural GLP-1 but lasts much longer in the blood, we can strongly activate the brain's satiety centers. This restores the signaling balance, allowing the person to feel full with smaller amounts of food. Downstream, this reduces the "reward" feeling associated with high-calorie foods and slows down the stomach’s movements. Unlike older weight-loss drugs that often worked like stimulants and had many side effects, GLP-1 therapies work with the body’s natural hormonal pathways, making them far more effective for long-term weight loss and overall metabolic health.
Disease:
Type 2 Diabetes
Prevalence:
589 million adults
Disease Mechanism:
Type 2 Diabetes is a systemic failure of glucose regulation characterized by insulin resistance and declining insulin production. Normally, gut hormones signal the pancreas to prepare for sugar, but this "incretin effect" falters in diabetics. As muscle and liver cells ignore insulin, blood sugar remains elevated. The pancreas overcompensates until its beta cells exhaust and fail. This crisis is compounded by a "leaky" liver overproducing glucose and a pancreas releasing excess glucagon. Ultimately, this breakdown in communication between the gut, liver, and pancreas creates a chronic cycle of high blood sugar that progressively damages the body's organs.
Why Target the drug target is used in this disease?
Activating the GLP-1 receptor is like "resetting" the body's glucose thermostat. It forces the pancreas to release insulin only when sugar is present, which prevents the dangerous blood sugar spikes and crashes seen with older medicines like sulfonylureas. It also tells the liver to stop producing unnecessary sugar by suppressing glucagon. Beyond just lowering sugar, these drugs protect the heart and kidneys, organs that are usually the first to suffer in diabetes. Because the mechanism is glucose-dependent, it has a very low risk of causing "hypoglycemia" (dangerously low blood sugar), which was a major limitation of older therapies.

.webp)