Disease Biology
Disease:
Alzheimer’s Disease (AD)
Prevalence:
Approximately 55 million people worldwide live with dementia, of which Alzheimer’s disease accounts for about 60–70% of cases.
Disease Mechanism:
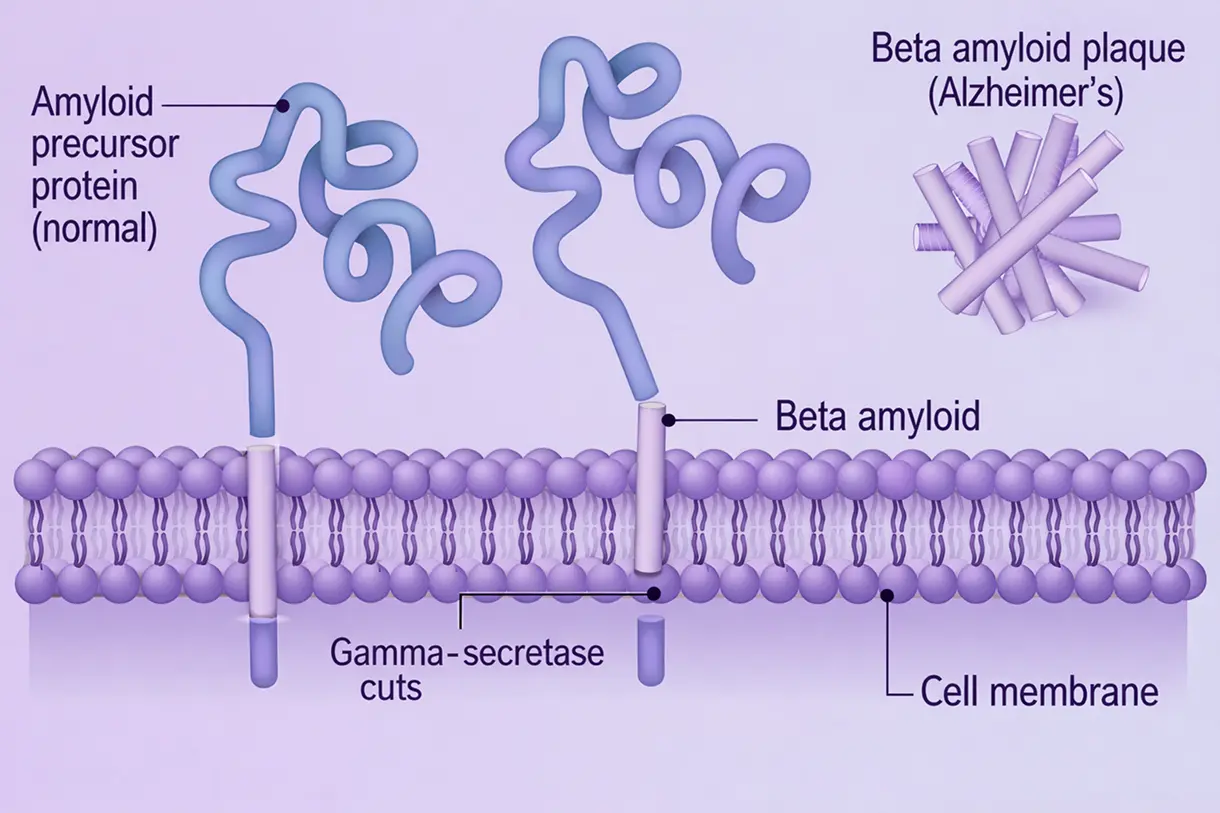
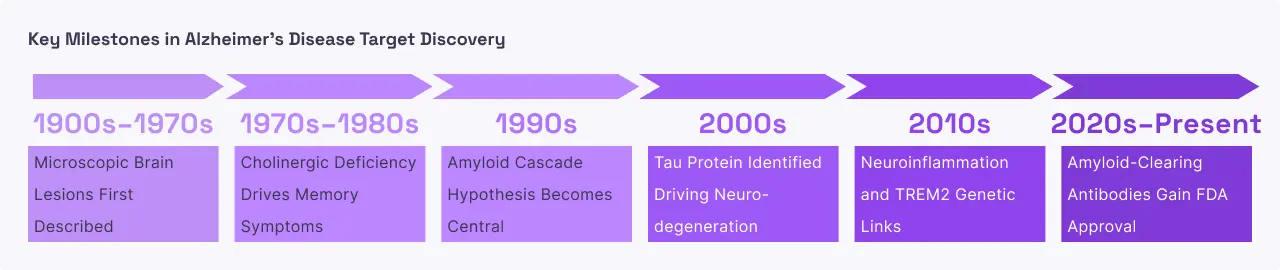
Alzheimer’s disease is a slow, progressive destruction of the brain that begins decades before the first sign of memory loss. The "Amyloid Cascade Hypothesis" suggests that the buildup of Amyloid Beta is the primary spark that ignites a larger fire. Once Aβ plaques reach a critical mass, they trigger a second protein called tau to malfunction and form tangles inside neurons. These tangles act like blockages in a railway, preventing nutrients from traveling through the cell. This dual attack plaques on the outside and tangles on the inside causes the brain’s immune cells (microglia) to become hyperactive and release inflammatory chemicals that damage healthy tissue. As connections between brain cells wither, the brain physically shrinks, beginning in memory centers like the hippocampus and eventually spreading to areas responsible for language, reasoning, and basic bodily functions.
Why target Amyloid Beta in Used in Alzheimer’s Disease?
Targeting Amyloid Beta is considered the "upstream" approach to stopping Alzheimer's. By removing Aβ plaques early in the disease process, scientists aim to prevent the subsequent "tau fire" and inflammation from ever starting. Modern monoclonal antibodies are engineered to recognize specific shapes of Aβ such as the toxic "protofibrils" and mark them for destruction by the brain's immune cells. Clinical data from recently approved therapies show that successfully clearing these plaques can "freeze" or significantly slow a patient's functional decline, allowing them to maintain independence for a longer period. Unlike older drugs that only temporarily masked symptoms, Aβ-targeted therapies aim to change the actual biology of the disease.
Disease:
Cerebral Amyloid Angiopathy (CAA)
Prevalence:
Present in approximately 30- 40% of older adults and up to 80- 90% of patients with Alzheimer’s disease.
Disease Mechanism:
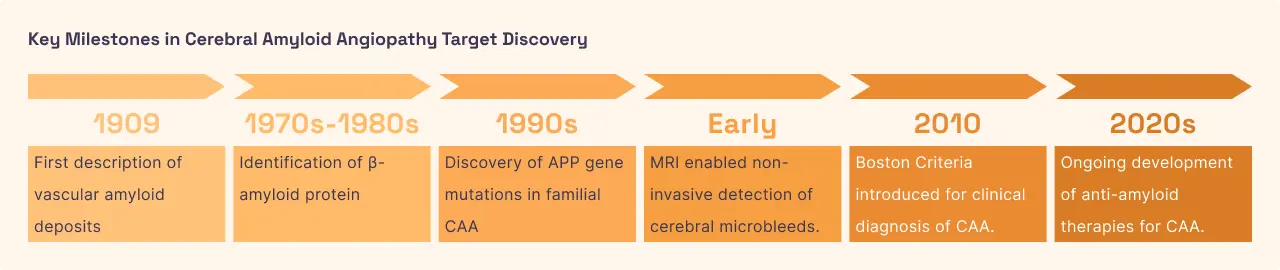
Cerebral Amyloid Angiopathy is a condition where Amyloid Beta deposits itself specifically within the walls of the brain’s small and medium-sized blood vessels rather than the brain tissue itself. Over time, these amyloid "bricks" replace the flexible muscular fibers of the vessel walls, making them brittle and fragile. This leads to two major problems: the vessels can become leaky, causing "microbleeds," or they can rupture entirely, leading to a major stroke. Furthermore, because the vessels are stiffened by amyloid, they cannot dilate properly to deliver more blood when the brain is working hard, leading to a "silent" lack of oxygen that contributes to cognitive decline and white matter damage.
Why target Amyloid Beta in Used in Cerebral Amyloid Angiopathy?
In CAA, Amyloid Beta is the direct cause of vascular fragility. By targeting the specific form of Aβ (Aβ40) that tends to congregate in blood vessels, researchers hope to restore the structural integrity of the brain's plumbing. While most current Aβ drugs were designed for Alzheimer's, their ability to clear vascular amyloid is a major area of active research. Reducing the "amyloid burden" in the vessel walls could potentially lower the risk of life-threatening brain bleeds and preserve the brain's blood supply. However, this must be done carefully, as rapid removal of amyloid from vessels can sometimes cause temporary swelling or leaks (known as ARIA).