Disease Biology
Disease:
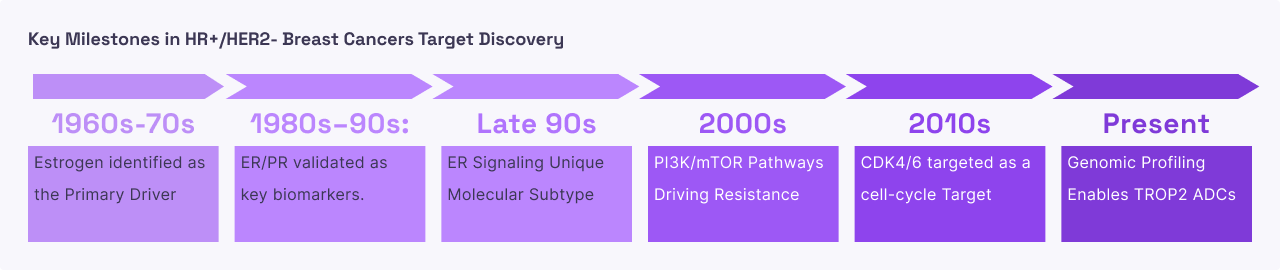
HR+/HER2- Breast Cancer
Prevalence:
1.6 million cases per year
Disease Mechanism:
Triple-negative breast cancer is an aggressive subtype of breast cancer defined by the absence of three common receptors that usually drive tumor growth: estrogen, progesterone, and HER2. Because it lacks these targets, traditional hormone therapies and HER2-targeted drugs do not work. At a molecular level, TNBC tumors often hijack growth pathways to compensate for these missing receptors, frequently overexpressing TROP2 to fuel their expansion. The disease progresses rapidly, often spreading to the lungs or brain more quickly than other breast cancer types. The lack of "anchors" for standard targeted drugs has historically left chemotherapy as the primary treatment option, which often leads to systemic toxicity and high rates of recurrence as the cancer develops resistance to general cell-killing agents.
Why Target the drug target is used in this disease?
TROP2 is overexpressed in nearly 90% of TNBC cases, making it a nearly universal target for this specific patient population. By using an Antibody-Drug Conjugate (ADC) directed at TROP2, doctors can deliver a "payload" of chemotherapy specifically to the cancer cells. Once the antibody attaches to TROP2 on the cell surface, the entire complex is swallowed by the cell, releasing the toxic medicine inside to destroy the cancer's DNA. This approach is significantly more effective than standard chemotherapy because it achieves a higher concentration of medicine within the tumor while reducing the amount of poison circulating in the patient’s bloodstream, thereby limiting side effects like severe nausea or extreme immune suppression.
Disease:
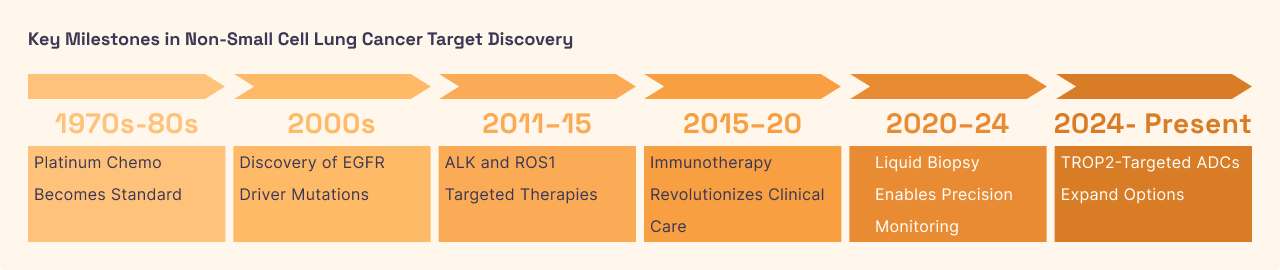
Non-Small Cell Lung Cancer (NSCLC)
Prevalence:
Approximately 2.1 million new cases worldwide in 2024
Disease Mechanism:
Non-small cell lung cancer accounts for the vast majority of all lung cancer cases. It begins when the epithelial cells lining the airways undergo genetic mutations, often triggered by environmental toxins like cigarette smoke or pollution. These cells lose their ability to self-destruct when damaged, leading to the formation of tumors that obstruct breathing and eventually hijack the blood supply to fuel further growth. As the disease progresses, the cancer cells often develop resistance to common treatments like "checkpoint inhibitors" (immunotherapy), creating a desperate need for new ways to identify and kill the mutated lung cells without destroying the delicate lung tissue required for oxygen exchange.
Why Target the drug target is used in this disease?
In NSCLC, high TROP2 levels are associated with poor prognosis and a higher likelihood of the cancer spreading. Unlike some targets that are only present in specific genetic mutations (like EGFR), TROP2 is widely present across many different types of lung cancer cells. This makes TROP2-targeted drugs a "broad-spectrum" option for patients who have failed other therapies. By modulating TROP2 via ADCs, clinicians can deliver a highly concentrated dose of medicine to the lung tumors. Newer TROP2 drugs are also designed with a "bystander effect," meaning the toxin can leak out of the primary target cell to kill neighboring cancer cells that might have lower levels of TROP2, ensuring a more thorough clearance of the tumor.