Disease Biology
Disease:
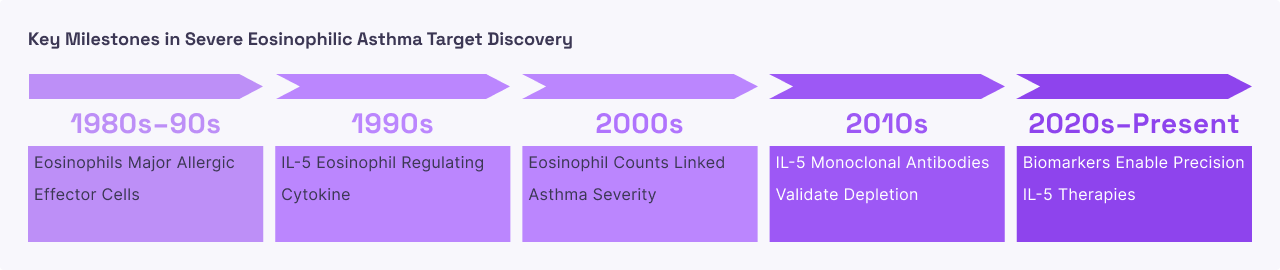
Severe Eosinophilic Asthma
Prevalence:
3.5 million people worldwide in 2023
Disease Mechanism:
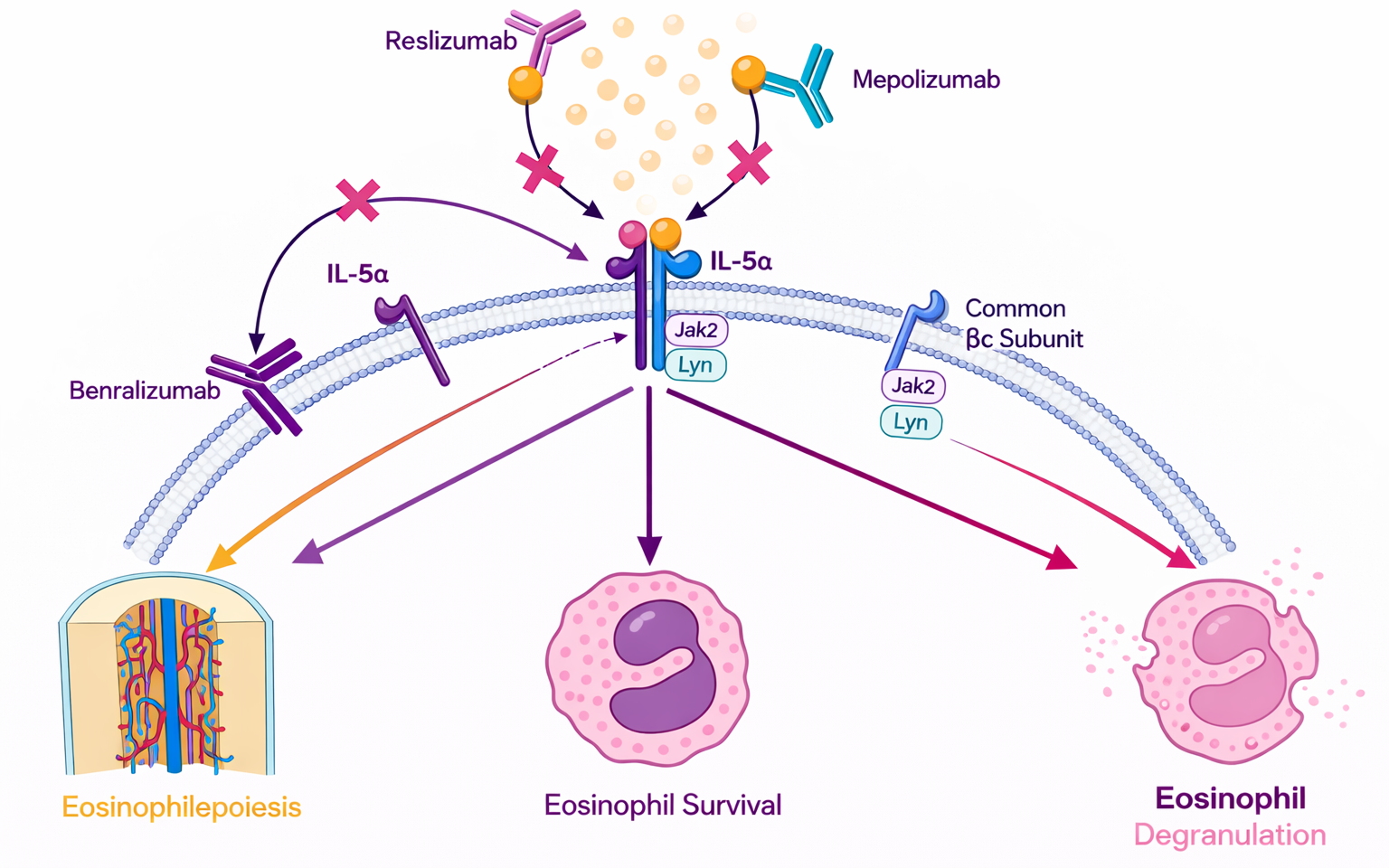
Severe eosinophilic asthma is a high-risk form of the disease that does not respond well to standard steroid inhalers. At a system level, the body’s immune system mistakenly identifies harmless environmental triggers as major threats, leading to a continuous overproduction of IL-5 . This causes a massive influx of eosinophils into the airways. Unlike "allergic" asthma, which is often triggered by specific external allergens, eosinophilic asthma is driven by this internal cellular imbalance . As these cells accumulate in the lung lining, they release "granules", microscopic chemical bombs that strip away the protective layer of the airways. Over time, this results in "airway remodeling," where the lungs become permanently scarred and thickened, making it increasingly difficult for the patient to breathe.
Why Target the drug target is used in this disease?
In severe eosinophilic asthma, IL-5 is the "master regulator" of the destructive inflammation. By introducing monoclonal antibodies that either bind to the IL-5 protein itself or block its receptor on the cell surface, the signal for eosinophil production is cut off. This leads to a dramatic drop in the number of eosinophils in both the blood and the lung tissue. Clinically, this "targeted depletion" is highly effective because it treats the underlying cause of the asthma flare-ups rather than just temporarily opening the airways . Patients often experience significantly fewer hospitalizations and a reduced need for oral steroids, which carry heavy long-term side effects like weight gain and bone loss.
Disease:
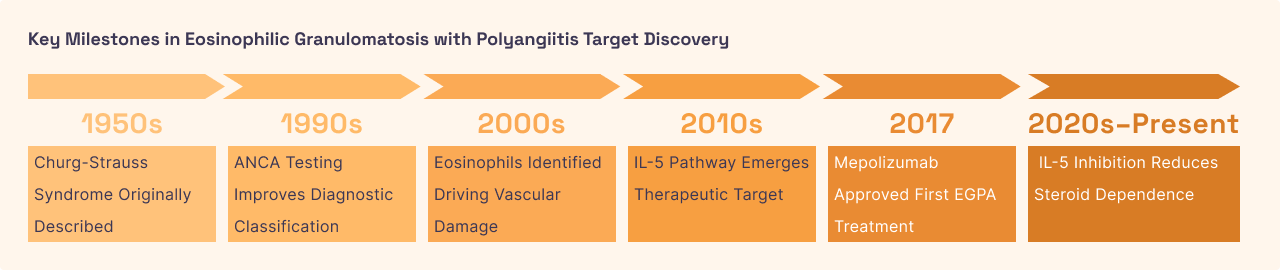
Eosinophilic Granulomatosis with Polyangiitis (EGPA)
Prevalence:
Approximately 2 to 5 per million people annually in 2024
Disease Mechanism:
EGPA, formerly known as Churg-Strauss Syndrome, is a rare autoimmune disease characterized by intense inflammation of small and medium-sized blood vessels (vasculitis). The disease typically progresses in three stages: it begins with severe asthma, followed by high levels of eosinophils in the blood, and finally results in widespread tissue damage. When eosinophils move from the blood into the vessel walls, they trigger inflammation that can restrict blood flow to vital organs, including the heart, kidneys, and nerves . Without treatment, this organ cross-talk can lead to permanent nerve damage or heart failure as the body’s own defense cells turn against its circulatory system.
Why Target the drug target is used in this disease?
Because EGPA is fundamentally driven by an extreme excess of eosinophils, blocking the IL-5 pathway is a logical and highly effective strategy. By neutralizing IL-5, the therapy forces the eosinophil count back down toward normal levels, which helps "cool off" the inflamed blood vessels . This allows the vessels to heal and restores proper blood flow to the organs. Compared to traditional high-dose chemotherapy or steroids used for vasculitis, IL-5 inhibitors provide a more "surgical" strike against the disease, offering better disease control with far fewer toxicities.